This is also the featured periodic table on the cover of our Periodic Table Journal available at Amazon. Visualize trends, 3D orbitals, isotopes, and mix compounds. It includes element names, symbols, groups, atomic numbers, and atomic masses. Interactive periodic table showing names, electrons, and oxidation states. It features our favorite color scheme of all the tables we’ve made. A screenshot is preferable to a picture of your laptop screen. This 118 element periodic table is a 1920×1080 HD wallpaper. Please do not ask for help acquiring, preparing, or handling illicit substances or for help with any activity that does not fall within the confines of whatever laws apply to your particular location.īonus points: If submitting a picture please make sure that it is clear. The elements in group 1 are known as the alkali metals, those in group 2. Groups are numbered 118 from left to right. Elements can be classified as metals, metalloids, and nonmetals, or as a main-group elements, transition metals, and inner transition metals. Any infractions will be met with a temporary ban at the first instance and a permanent ban if there is another. Elements in the same group of the periodic table have similar chemical properties. For main-group elements, those categorized in groups 1, 2, and 13-18, form ions they lose the same number of electrons as the corresponding group number to which they fall under. The periodic table of elements is useful in determining the charges on simple monoatomic ions. It is also important that you describe the specific part of the problem you are struggling with. Useful Relationships from the Periodic Table. It is OK if you are a little (or a lot!) stuck, we just want to see that you have made an effort. 
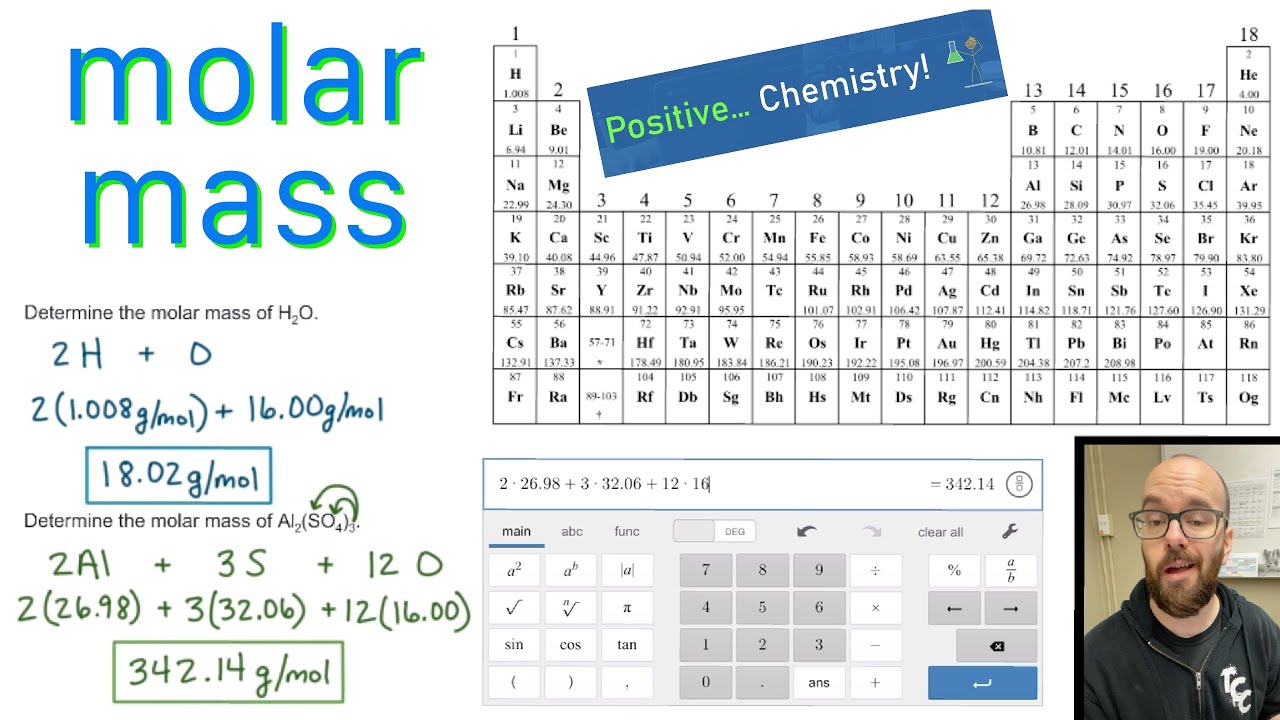
Please complete any questions as much as you can before posting. We will not do your homework for you, so don't ask. The next element is the calcium.Please flair yourself and read over the rules below before posting. A number of neutrons are 14 and it belongs to the third period now. Atomic number is 13, has 13 plotons, 13 electrons, it has atomic mass of 26.982 and it has around atomic mass 27. You will also get the HD images of the Periodic table (for FREE). You will get the detailed information about the periodic table which will convert a newbie into pro. For calculations, use the given molar mass on the periodic table without rounding. You can effortlessly find every single detail about the elements from this single Interactive Periodic table. The next element is the alminium alminium. Thermal Conductivity0.1805 W/ (m K) Specific Heat 14300 J/ (kg K) Heat Fusion 0.558 kJ/mol. Re: Rounding molar mass on the periodic table. We have to subtract mass number from protons, so it comes to be 6 and it belongs to the second period. Next, using the periodic table, find the atomic mass in g/mol of each element (the molar mass of an element is equal to its atomic mass): Molar Mass (g/mol) H (Hydrogen) 1.00794: S (Sulphur/Sulfur) 32.065: O (Oxygen) 15.9994: 3. One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. Rounded off mass number is 12 point, so its number of neutrons are. Molar mass is the mass (in atomic mass units) of one mole of a of a substance. It has 6 protons 6 electrons, as number is 12.0 11. Is the son has a symbol c: its atomic number is 6. As a result, this chart actually displays the mass number of a specific isotope for each element. Atomic mass of all elements (along with the rounded off values) is mentioned inside the chart below. The atomic weights listed on this Table of Elements have been rounded to the nearest whole number. The number of protons, so it comes to be 2, which belongs to the first period now the next given element is the next given element. Atomic mass of all units (along with the rounded out values) is mentioned in the chart below. The first element is the helium first element is the helium, so the simple used for helium is its atomic number, is 2 number of protons are 2? Electrons are 2? Mass number is 4.0 2 rounded, as 4 number of neutrons are a number of mass number minus. Then their atomic number than the number of protons present in it than the number of electrons present in it than the atomic mass number than the rounded rounded mass number than the neutrons present in it. The elements that are given in the question that must be written here is the symbols that used to designate them. 
Number of protons so the elements so the given elements are firstly here. In Chapter 1, you learned that 1 mole of any substance is equivalent to 6.02 X 10 23 molecules of that substance. Check the periodic table for the correct answer Molar Mass. Boron exists as a mixture that is 19.9 10 B and 80.1 11 B. There are certain elements that are given in the question and we have to find their symbols. Try to work out the atomic mass for boron. The elements are arranged according to their groups and periods according to their chemical properties. Hello students- let's solve this question, this question is based to the periodic table in periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
